ALS Treatment Pathways: A Comprehensive Guide
℞ Approved Therapies: Disease Modifiers
Riluzole
Availability: A generic drug widely available in many countries under brand names like Rilutek, Tiglutik, and Exservan.
Approval: First approved by the U.S. Food and Drug Administration (FDA) in 1995 and has long been the only approved disease-modifying treatment in many regions, including the European Union (EU).
Mechanism: Inhibits glutamate release, a neurotransmitter that can be toxic to motor neurons at high levels.

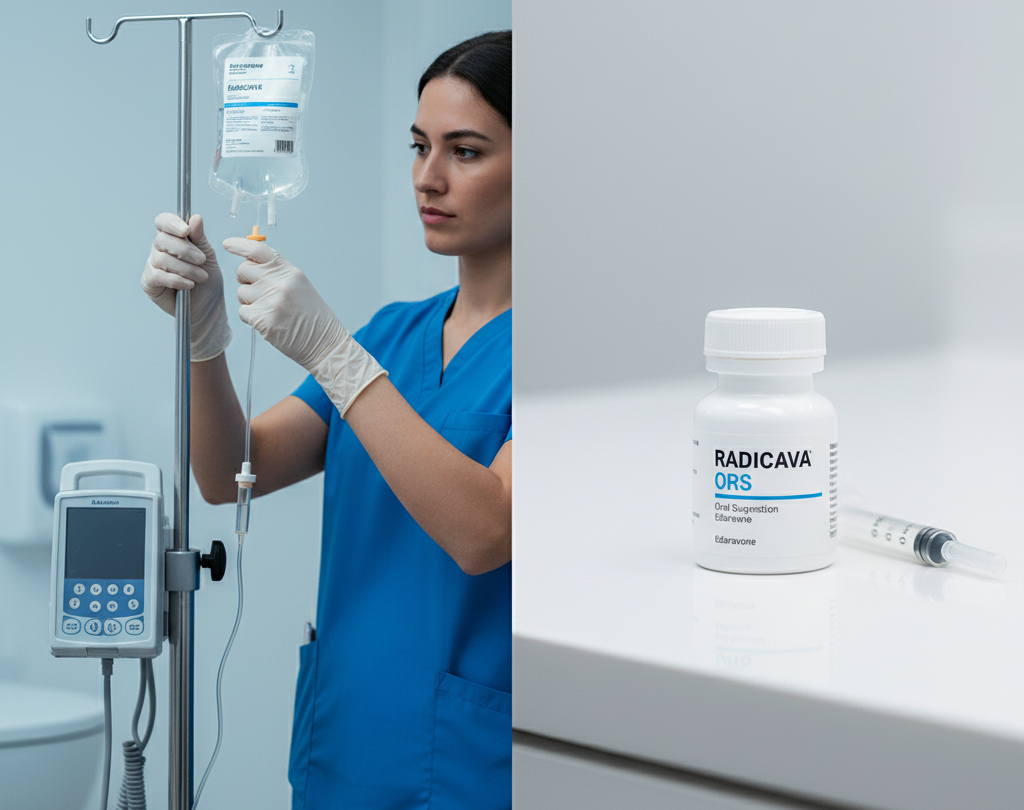
Edaravone (Radicava)
Availability: Approved in the U.S., Canada, Japan, Switzerland, China, South Korea, and Indonesia, with both intravenous (IV) and oral formulations. The oral version, Radicava ORS, was approved in the U.S. in 2022.
Mechanism: A free radical scavenger and antioxidant that reduces oxidative stress, which is a key factor in ALS.
Tofersen (Qalsody)
Availability: An expensive, specialized therapy with limited availability for a rare, specific form of ALS.
U.S.: Received accelerated FDA approval in 2023 for adults with a superoxide dismutase 1 (SOD1) gene mutation.
Europe: Gained European Commission approval in May 2024 for SOD1-ALS.
Mechanism: An antisense oligonucleotide that targets the messenger RNA (mRNA) of the SOD1 gene to reduce the production of the toxic SOD1 protein.

Symptomatic Treatments: Quality of Life
These medications target specific symptoms to help manage the daily challenges of ALS.
Nuedexta (Dextromethorphan/Quinidine)
FDA-approved for **pseudobulbar affect (PBA)**, managing uncontrolled emotional outbursts such as laughing or crying.
Side effects: dizziness, diarrhea, fatigue.
Baclofen and Tizanidine
Help manage **muscle spasticity and stiffness**. Baclofen acts as a muscle relaxant; Tizanidine reduces muscle tone.
Side effects: drowsiness, dizziness, muscle weakness.
Gabapentin and Pregabalin
Often prescribed to manage **neuropathic pain and discomfort** by modulating nerve signaling.
Side effects: dizziness, fatigue, weight gain.
Off-Label & Experimental Treatments: The Frontier
This section explores promising compounds and approaches currently under investigation or used off-label.
TUDCA is a bile acid derivative currently under investigation for ALS treatment. It may protect motor neurons by reducing apoptosis and oxidative stress. Early clinical trials suggest potential benefits in slowing disease progression. It is not yet FDA-approved for ALS.
Clenbuterol is a beta-2 adrenergic agonist primarily used for respiratory conditions. It has been explored off-label for ALS due to its potential anabolic and neuroprotective effects. However, evidence supporting its effectiveness in ALS remains limited, and it is not FDA-approved for ALS treatment. Side effects include tremors, palpitations, and anxiety.
Lunasin is a peptide derived from soybeans, proposed as a potential ALS treatment due to anti-inflammatory and antioxidant properties. Limited anecdotal reports and small studies have suggested possible benefits, but robust clinical data are lacking. Lunasin is not FDA-approved for ALS.
Sodium phenylbutyrate, a drug primarily used in urea cycle disorders, has been investigated for ALS due to its potential neuroprotective effects. It is one component of the FDA-approved ALS medication Relyvrio. However, as a standalone treatment, it remains experimental and not specifically FDA-approved for ALS.
Ibudilast is an anti-inflammatory and neuroprotective drug being studied for ALS. Early clinical trials have indicated potential benefits in slowing disease progression, but it remains experimental and is not FDA-approved for ALS.
An oral tyrosine kinase inhibitor originally developed for veterinary use and certain human cancers, masitinib has shown promise in clinical trials for ALS. Early studies suggest masitinib might slow disease progression in certain ALS patient subgroups. However, it remains experimental and is not yet officially approved for ALS treatment.
These drugs are fast skeletal muscle troponin activators designed to improve muscle function and strength. Clinical trials have explored their potential to enhance muscle performance in ALS patients. Although initial studies have provided some encouraging results, neither tirasemtiv nor reldesemtiv has received regulatory approval for ALS, and further research is ongoing.
Stem cell treatments have been extensively investigated as potential therapeutic options for ALS. Researchers hope that stem cells could replace damaged motor neurons or provide supportive factors to slow neuron degeneration. Clinical trials involving stem cell transplantation have demonstrated safety and tolerability, but efficacy results have been mixed and inconclusive. Currently, stem cell therapies remain experimental and are not approved as standard treatment for ALS.
© 2025
All Rights Reserved.
